MIT researchers have developed an implantable device that encapsulates insulin-producing islet cells and includes an on-board oxygen generator, potentially offering long-term diabetes control without immune suppression.
For millions of people living with type 1 diabetes, daily life revolves around carefully monitoring blood sugar levels and administering insulin injections multiple times per day. This constant vigilance is necessary to prevent dangerous blood sugar spikes that can lead to serious complications. Now, MIT researchers are developing a revolutionary implantable device that could transform diabetes treatment by eliminating the need for daily injections while avoiding the immune suppression required by current cell therapies.
A New Approach to Cell Therapy
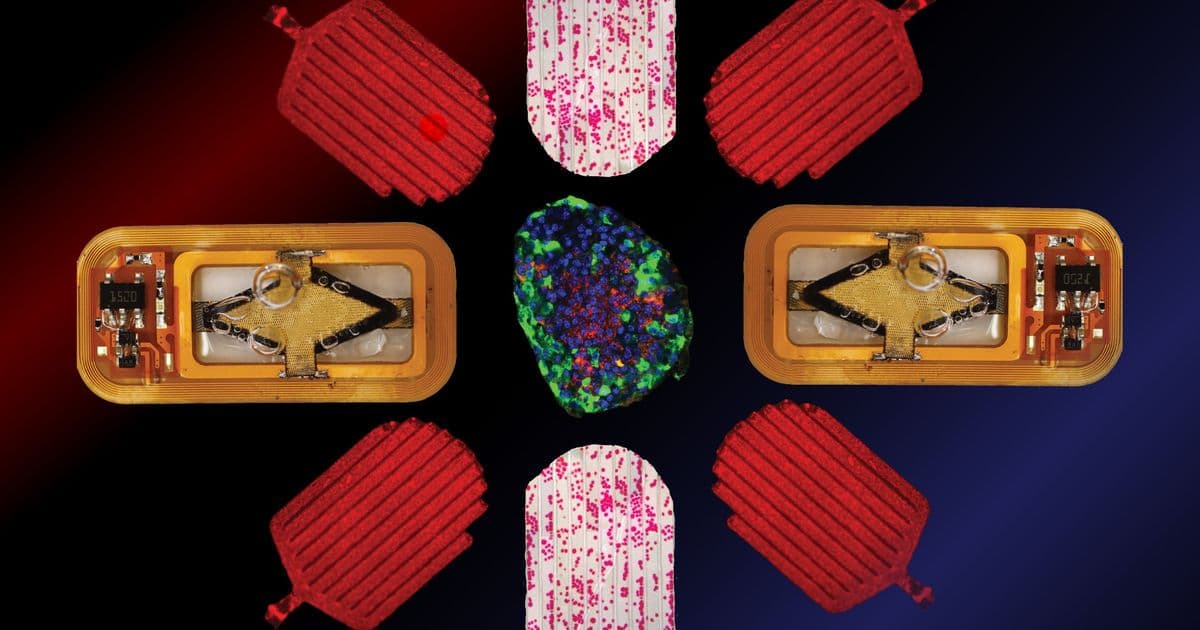
The device, detailed in a recent study published in the journal Device, encapsulates insulin-producing pancreatic islet cells within a protective coating. This encapsulation serves a dual purpose: it shields the foreign cells from immune rejection while allowing them to function normally. The key innovation, however, lies in the device's built-in oxygen generator, which keeps the encapsulated cells healthy and productive for extended periods.
"Islet cell therapy can be a transformative treatment for patients. However, current methods also require immune suppression, which for some people can be really debilitating," explains Daniel Anderson, a professor in MIT's Department of Chemical Engineering and senior author of the study. "Our goal is to find a way to give patients the benefit of cell therapy without the need for immune suppression."
How It Works
The oxygen generator at the heart of the device uses a proton-exchange membrane to split water vapor—abundant in the body—into hydrogen and oxygen. The hydrogen harmlessly diffuses away, while oxygen is stored and delivered to the islet cells through a thin, oxygen-permeable membrane. This self-sustaining system means the device doesn't require replacement or refilling, making it ideal for long-term implantation.
Power is delivered wirelessly through an external antenna placed on the skin, which transfers energy to the device. By optimizing the device's circuitry, the researchers significantly increased the power reaching the oxygen-generating system, allowing it to produce more oxygen and support healthier, more productive islet cells.
Promising Results in Animal Studies
In tests conducted on mice and rats, the improved device demonstrated remarkable durability and effectiveness. The encapsulated islet cells survived for at least 90 days after implantation—a substantial improvement over the one-month lifespan achieved in earlier versions. During this three-month period, the cells produced enough insulin to maintain healthy blood sugar levels in the animals.
The researchers also tested the device with islet cells derived from induced pluripotent stem cells, which could potentially provide an unlimited supply of cells for any patient who needs them. While these stem cell-derived islets didn't fully reverse diabetes in the test animals, they did achieve significant blood sugar control.
"We're hoping that in the future, if we can give the cells a little bit longer to fully mature, that they'll secrete even more insulin to better regulate diabetes in the animals," says Matthew Bochenek, a former MIT postdoc and lead author of the study.
Beyond Diabetes Treatment
The implications of this technology extend far beyond diabetes management. The researchers envision using similar devices to deliver cells that produce other therapeutic proteins, such as antibodies, enzymes, or clotting factors. This could revolutionize treatment for numerous conditions that currently require repeated, lengthy infusions of protein-based therapies.
"We think that these technologies could provide a long-term way to treat human disease by making drugs in the body instead of outside of the body," Anderson notes. "There are many protein therapies where patients must receive repeated, lengthy infusions. We think it may be possible to create a device that could continuously create protein therapeutics on demand and as needed by the patient."
The Road Ahead
While the 90-day results are promising, the research team aims to extend the device's lifespan even further—potentially up to two years or more. They're also working to optimize the maturation process of the islet cells to improve their insulin production capabilities.
The research, funded by organizations including Breakthrough TID, the Leona M. and Harry B. Helmsley Charitable Trust, and the National Institutes of Health, represents a significant step toward making cell therapy a practical reality for diabetes patients without the drawbacks of immune suppression.
For the millions of people who must carefully balance their blood sugar levels every day, this technology offers hope for a future where managing diabetes becomes as simple as having a small, self-sustaining device implanted in the body—one that works quietly and continuously to maintain healthy blood sugar levels without daily intervention.
The development of this implantable device exemplifies how engineering innovation can address longstanding medical challenges, potentially transforming the lives of patients with chronic conditions that require ongoing therapeutic intervention.
Comments
Please log in or register to join the discussion