MIT chemists have created a precision technique that allows scientists to seamlessly relocate alcohol functional groups from one spot on a molecule to a neighboring site, bypassing the need to rebuild entire structures.
MIT chemists have developed a groundbreaking molecular editing tool that allows researchers to relocate alcohol functional groups within organic molecules with unprecedented precision. This innovation, recently published in Nature, represents a significant leap forward in chemical synthesis, potentially transforming how scientists approach drug discovery, materials science, and agrochemical development.
The Challenge of Molecular Fine-Tuning
In the world of molecular design, even the smallest structural change can dramatically alter a molecule's function. Historically, making these adjustments required researchers to completely rebuild the target molecule from scratch—a process that was both time-consuming and expensive. As Professor Alison Wendlandt, who led the research, explains, it was like "tearing down a house just to move a lamp."
This limitation has been a persistent bottleneck in fields ranging from pharmaceuticals to materials science. The inability to make precise, localized modifications to complex molecules has forced researchers to choose between accepting suboptimal structures or investing enormous resources in complete resynthesis.
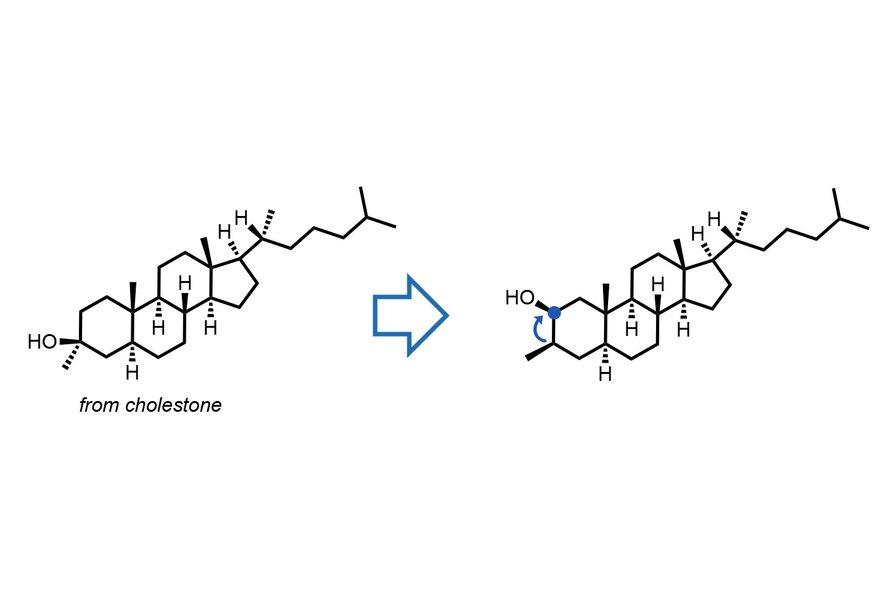
The Breakthrough: Light-Activated Molecular Migration
The MIT team's solution leverages a light-sensitive catalyst called decatungstate to trigger a highly controlled migration of alcohol groups. This process allows the alcohol functional group to move from one position to a neighboring site on the same molecule without disrupting the overall structure.
What makes this technique particularly remarkable is its predictability. The reaction maintains the molecule's precise 3D shape and orientation throughout the migration process, ensuring that the structural integrity remains intact. This level of control is crucial for applications in drug development, where even minor changes in molecular geometry can affect a drug's efficacy and safety profile.
Late-Stage Editing Capabilities
Perhaps the most exciting aspect of this discovery is its ability to work on complex, nearly finished molecular structures. Traditional synthesis methods often require modifications to be made early in the process, before the molecule becomes too complex. The MIT team's technique, however, is gentle enough to be applied to late-stage drug candidates, opening up new possibilities for fine-tuning molecules that are already close to their final form.
Qian Xu, co-first author of the paper and a postdoc in the Wendlandt Group, emphasizes the significance of this capability: "With predictable stereo- and regioselectivity and late-stage operability, it presents an enticing chance to modify natural products and drug molecules through 'editing.'"
Collaborative Innovation
The development of this molecular editing tool was the result of a multi-year collaboration with Bristol Myers Squibb, highlighting the importance of industry-academia partnerships in advancing chemical research. The team included MIT contributors Yichen Nie (co-lead author and graduate student), Ronghua Zhang (recent postdoc), and Jeremiah A. Johnson (professor of chemistry), as well as researchers from the University of Groningen and Actithera.
Implications for Drug Discovery and Beyond
This precision editing tool has the potential to dramatically improve the efficiency of molecular design campaigns across multiple industries. In pharmaceuticals, it could accelerate the development of new drugs by allowing researchers to make subtle but crucial modifications to existing candidates without starting from scratch. For materials science, it opens up new pathways to create challenging molecular architectures and oxygenation patterns that were previously difficult or impossible to achieve.
The technique's ability to implement subtle structural tweaks without the waste of complete resynthesis addresses a primary hurdle that has long plagued the field. By providing a more efficient way to explore chemical space and optimize molecular properties, this tool could help bring new therapies to patients faster and at lower cost.

The Future of Molecular Design
When combined with existing chemical methods, this alcohol migration strategy provides chemists with unprecedented control over molecular architecture. It represents a shift from the traditional approach of building molecules piece by piece to a more flexible paradigm where existing structures can be edited and refined like text in a document.
As Professor Wendlandt's work demonstrates, the future of molecular design lies in developing tools that offer both precision and flexibility. This breakthrough not only solves a long-standing challenge in chemical synthesis but also opens up entirely new possibilities for how we approach molecular engineering.
The ability to "edit" molecules with such precision brings us closer to a world where the development of new pharmaceuticals, materials, and agrochemicals can be achieved with greater efficiency, less waste, and more creative freedom than ever before.

Comments
Please log in or register to join the discussion