MIT researchers discover that cell membrane composition directly influences the activity of EGFR receptors, potentially explaining how cancer cells maintain uncontrolled growth.
MIT chemists have uncovered a surprising new role for cell membranes in cellular signaling, challenging the long-held view that these structures merely provide passive support for embedded proteins. The research reveals that membrane composition can actively regulate the function of EGFR, a receptor protein often implicated in cancer growth.
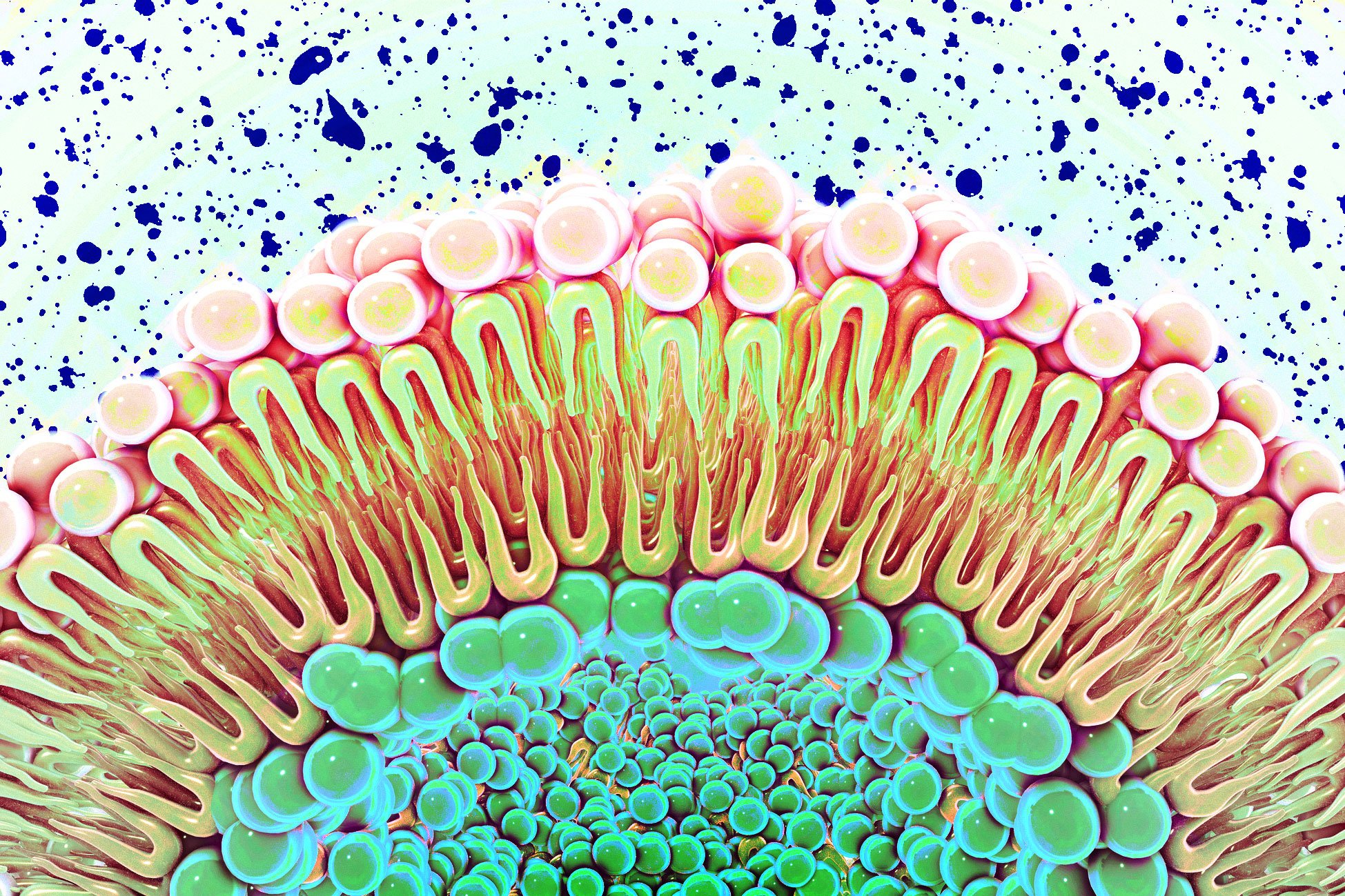
For decades, cell membranes were understood primarily as structural barriers—lipid bilayers that compartmentalize cells and provide a scaffold for protein receptors. However, mounting evidence suggested these membranes might play a more dynamic role in cellular function. The new MIT study provides compelling evidence that membrane composition directly influences how receptors behave, particularly those involved in cell proliferation.
The EGFR Connection to Cancer
The epidermal growth factor receptor (EGFR) sits on the surface of cells lining organs and body surfaces, where it normally helps regulate cell growth and division. When functioning properly, EGFR responds to specific signals by triggering controlled cell proliferation. However, in many cancers—especially lung cancer and glioblastoma—EGFR becomes overactive, driving uncontrolled cell division.
What makes this discovery particularly significant is the mechanism by which membrane composition affects EGFR activity. The researchers found that when cell membranes contain elevated levels of negatively charged lipids, EGFR becomes locked into an active state. This means the receptor continuously signals cells to grow, even without the presence of growth factors that would normally activate it.
Technical Innovation Enables Discovery
Studying how membrane composition affects full-length receptor proteins has historically been extremely challenging. Traditional methods struggled to create membranes containing proteins that span the entire membrane structure, making it difficult to observe how signals propagate across the receptor.
To overcome this limitation, the MIT team employed nanodiscs—specially engineered membrane structures that self-assemble and can incorporate full-length receptors. This approach allowed researchers to study the complete receptor, from the portion that extends outside the cell through the membrane-spanning region to the intracellular signaling domain.
Using single molecule FRET (fluorescence resonance energy transfer), the team could measure how different parts of the EGFR protein move relative to each other under various conditions. This technique involves labeling different protein regions with fluorescent tags and measuring how quickly energy transfers between them, providing precise distance measurements at the molecular level.
The Charge Effect
The experiments revealed a critical threshold effect. When negatively charged lipids comprised 15 to 30 percent of the membrane—typical physiological levels—EGFR functioned normally. However, when these charged lipids reached 60 percent of membrane composition, the receptor became permanently activated.
This finding helps explain a puzzling observation in cancer biology: many cancer cells exhibit elevated levels of negatively charged membrane lipids. The study suggests this isn't merely a side effect of cancer but may actively contribute to maintaining the uncontrolled growth characteristic of malignant cells.
"If the membrane has high levels of negatively charged lipids, then it's always in that open conformation," explains Gabriela Schlau-Cohen, the study's senior author. "It doesn't matter if ligand is bound or unbound. It's always in the conformation that's telling the cell to grow, not just when EGF binds."
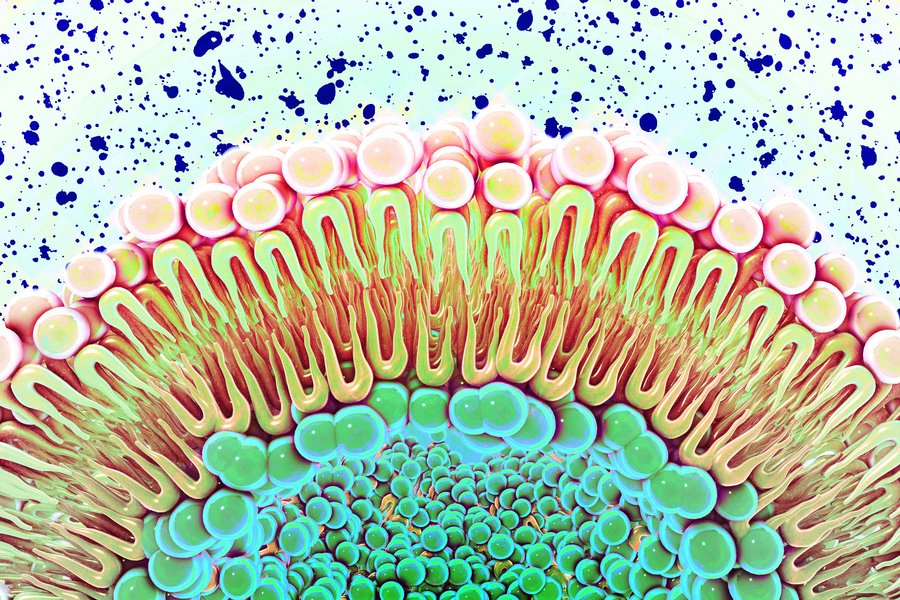
Cholesterol's Surprising Role
The research also uncovered an unexpected effect of cholesterol on EGFR signaling. When the team created nanodiscs with elevated cholesterol levels, the membranes became more rigid, and this increased rigidity suppressed EGFR signaling. This finding adds another layer of complexity to how membrane properties influence cellular behavior.
Implications for Cancer Treatment
These discoveries open new avenues for cancer therapy development. Rather than targeting the receptor protein itself, future treatments might focus on neutralizing the negative charge in cancer cell membranes or modulating membrane rigidity. Such approaches could potentially turn down EGFR signaling without directly interfering with the receptor's structure.
The study, published in the journal eLife, represents a significant shift in our understanding of cell membrane function. Far from being passive structural elements, membranes appear to actively participate in regulating critical cellular processes through their physical and chemical properties.
This research was funded by the National Institutes of Health and MIT's Department of Chemistry, highlighting the importance of fundamental scientific investigation in uncovering new therapeutic targets for complex diseases like cancer.

The findings underscore a broader principle in biology: cellular components often have multiple, interconnected roles that only become apparent through careful, innovative experimentation. As our tools for studying cellular systems improve, we continue to discover that biological processes are even more sophisticated and interconnected than previously imagined.
Comments
Please log in or register to join the discussion